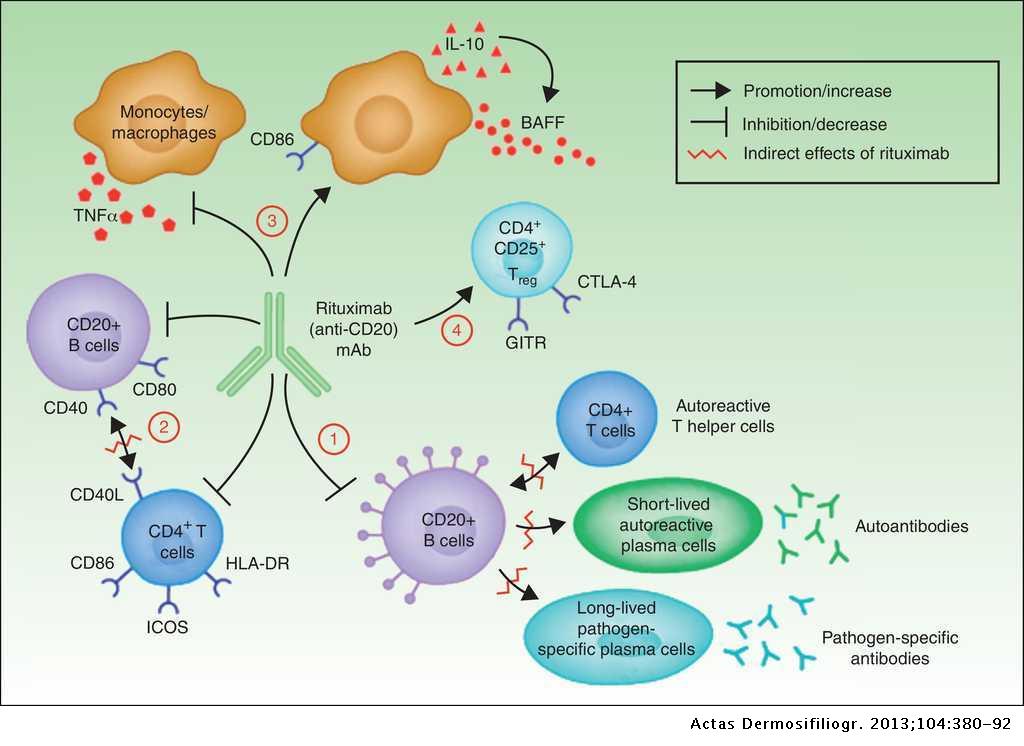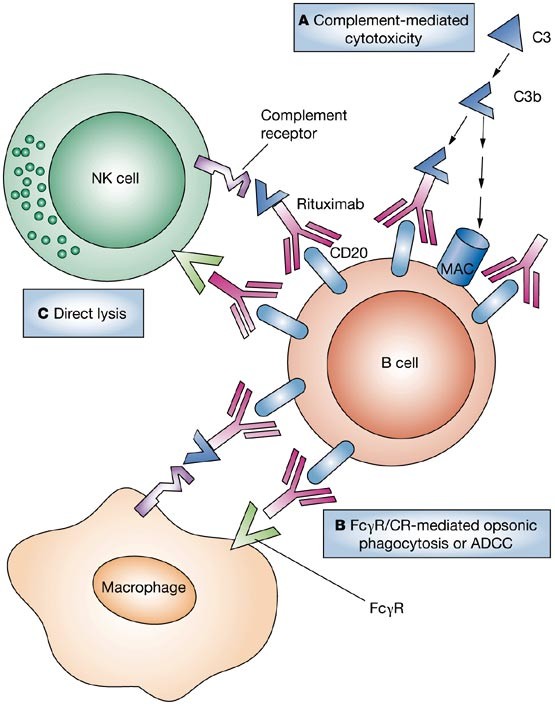
Drug Insight: the mechanism of action of rituximab in autoimmune disease—the immune complex decoy hypothesis | Nature Reviews Rheumatology

KISA ÜRÜN BĐLGĐSĐ. MABTHERA 500 mg/50 ml i.v. infüzyon için konsantre solüsyon içeren flakon - PDF Ücretsiz indirin

Romatoid artrit hastalarında Rituksimab (Mabthera) tedavisi ne zaman kullanılır? Bu tedaviyi alan hastalar nelere dikkat etmelidir? - Romatizma TV

Comparative Nonclinical Assessments of the Proposed Biosimilar PF-05280586 and Rituximab (MabThera®) - Anne M. Ryan, Sharon A. Sokolowski, Chee-Keng Ng, Norimitsu Shirai, Mark Collinge, Amy C. Shen, Joshua Arrington, Zaher Radi, Thomas

Romatoid artrit hastalarında Rituksimab (Mabthera) tedavisi ne zaman kullanılır? Bu tedaviyi alan hastalar nelere dikkat etmelidir? - Romatizma TV

Romatoid artrit hastalarında Rituksimab (Mabthera) tedavisi ne zaman kullanılır? Bu tedaviyi alan hastalar nelere dikkat etmelidir? - Romatizma TV

Rituximab Serum Concentrations and Anti-Rituximab Antibodies During B-Cell Depletion Therapy for Myalgic Encephalopathy/Chronic Fatigue Syndrome - ScienceDirect

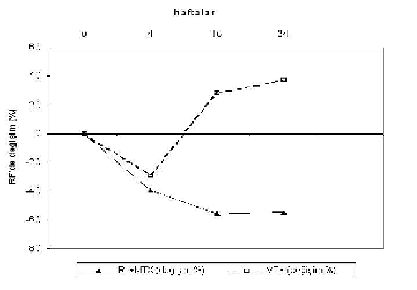
Serum rituximab concentrations in patients receiving chemoimmunotherapy | Download Scientific Diagram

Pharmacokinetics of serum rituximab in cynomolgus monkeys. PK analysis... | Download Scientific Diagram

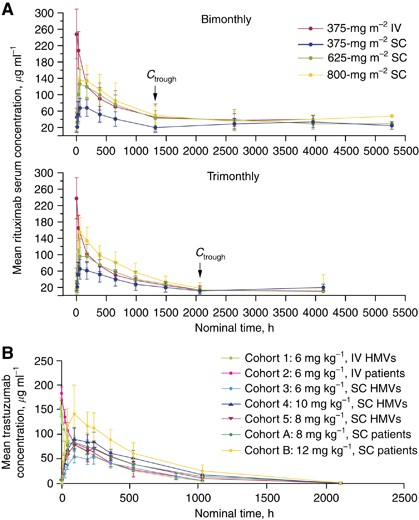
A) Mean serum concentration of rituximab over time by administration... | Download Scientific Diagram